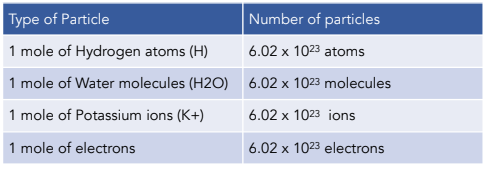
Calculate the value of Avogadro number from the internuclear distance of adjacent ions in NaCl , 0.282 nm and the density of solid NaCl is 2.17 × 10^3 kg/m^3 .A unit cell

Converting Between Particles and Moles Using Avogadro's Number and Conversion Factors – Science and Joe

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5
![How to calculate number of particles| Avogadro's constant| Mole and Chemical Calculation [Online Video] – O Level Secondary Chemistry Tuition How to calculate number of particles| Avogadro's constant| Mole and Chemical Calculation [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2020/12/thumb-insta-2.jpg)
How to calculate number of particles| Avogadro's constant| Mole and Chemical Calculation [Online Video] – O Level Secondary Chemistry Tuition

Physical Chemistry #1: Relative Mass, the Mole and Avogadro's Constant (Slides & Student Led Tasks) | Teaching Resources
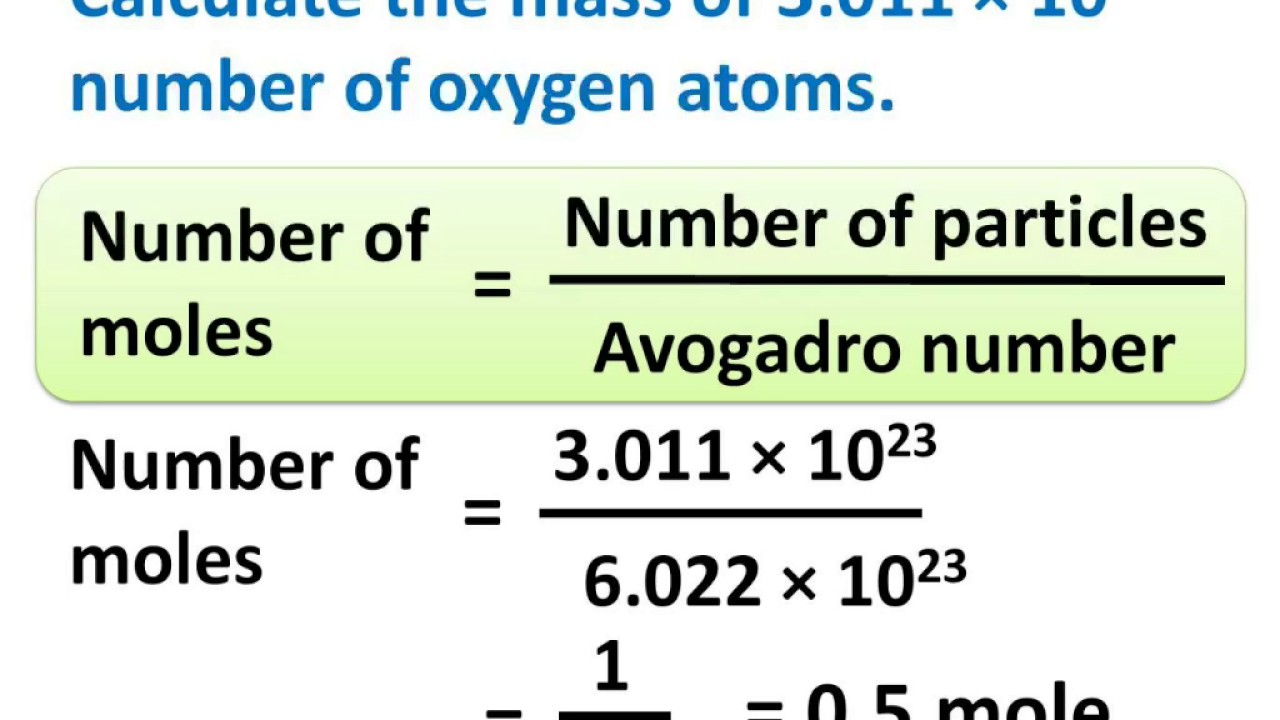


.PNG)