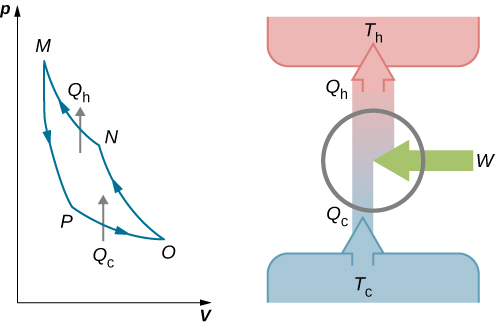
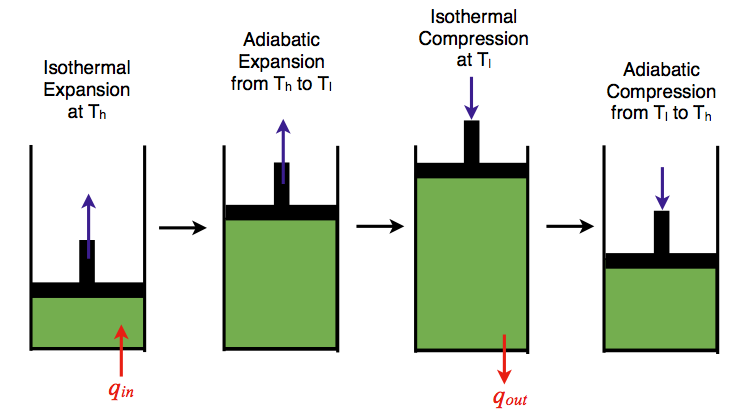
Carnot heat engine: a) basic schematic diagram, b) representation of... | Download Scientific Diagram
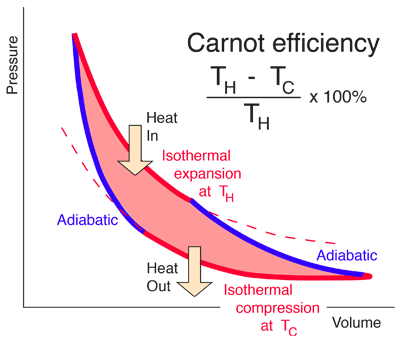
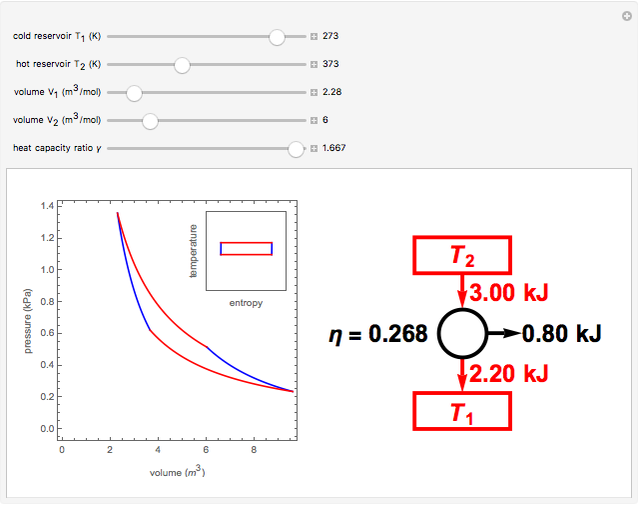
How to explain the Carnot cycle from thermodynamics? What are the efficiency, heat, and work calculations - Quora

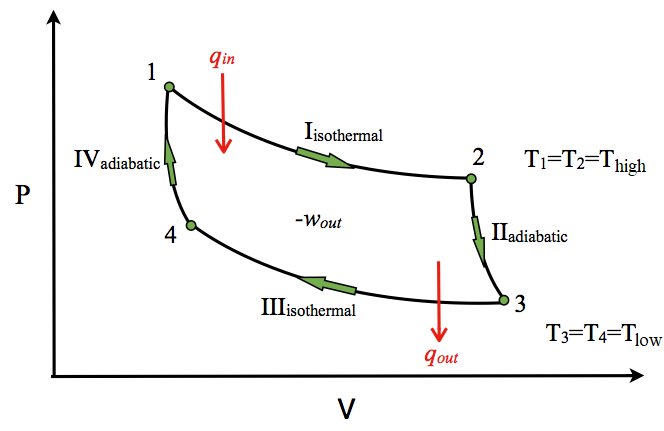
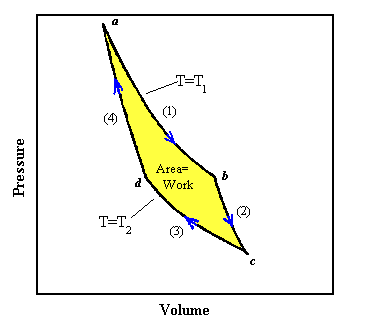
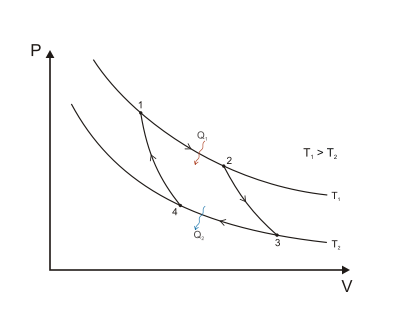
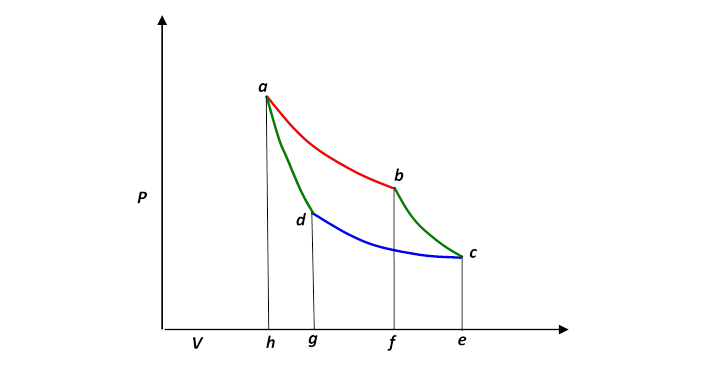
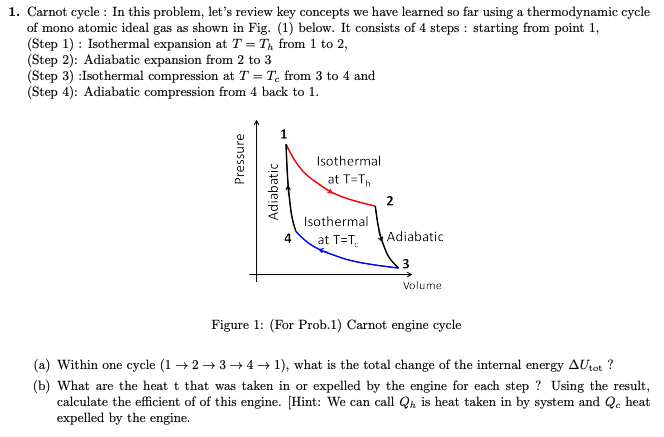
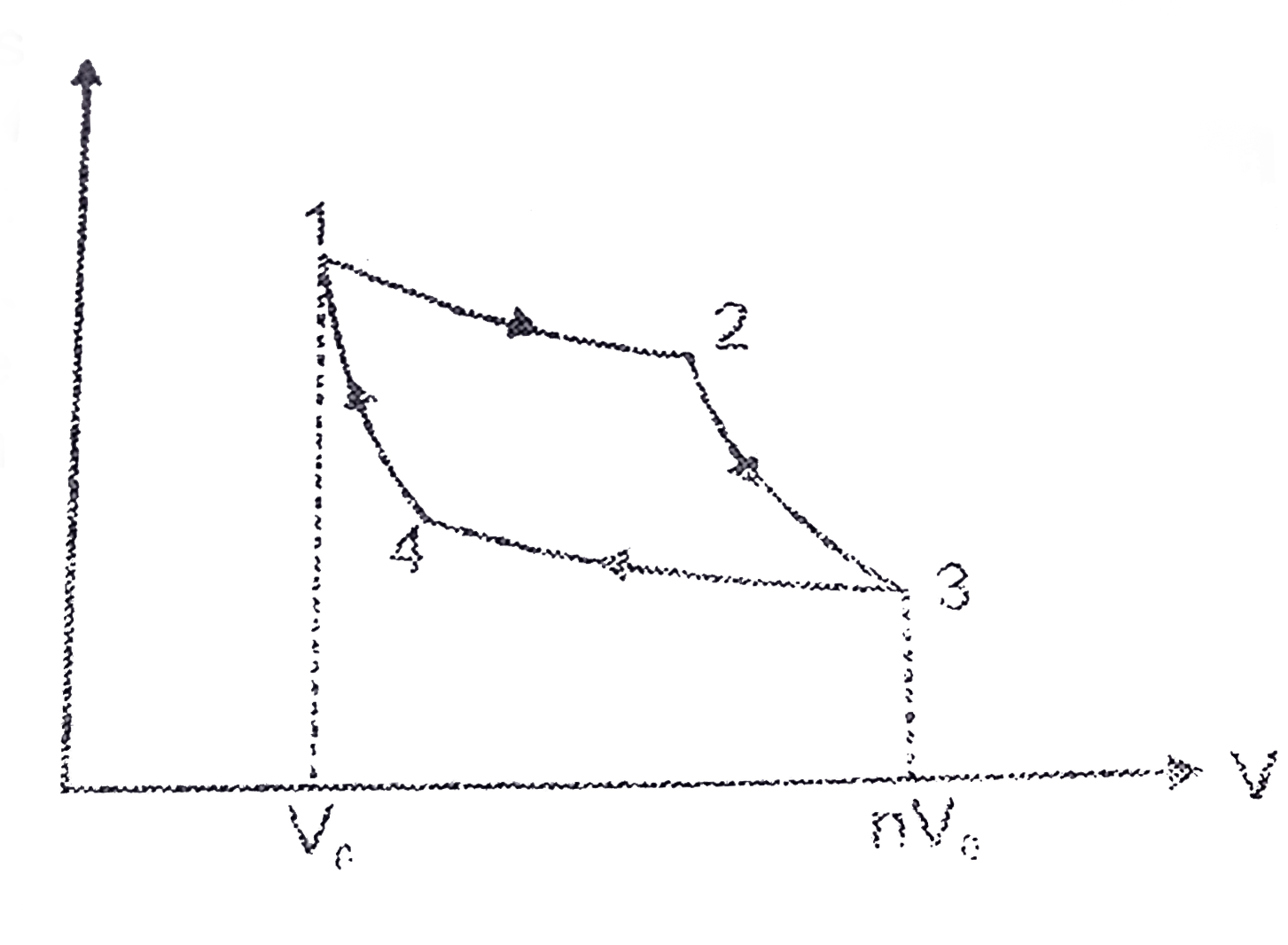
The p − V indicator diagram of a Carnot cycle 1-2-3-4, where T C < T H . | Download Scientific Diagram

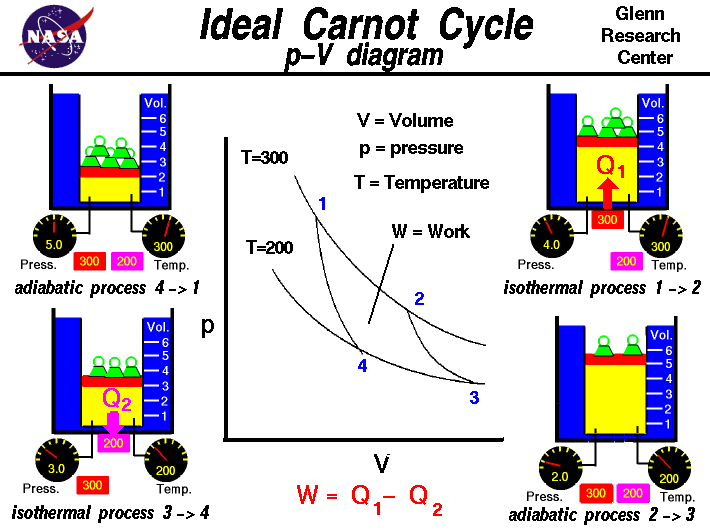
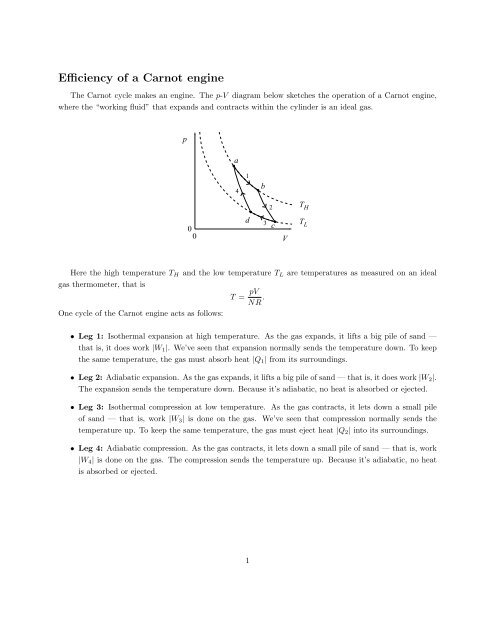
Carnot Heat Engines, Efficiency, Refrigerators, Pumps, Entropy, Thermodynamics - Second Law, Physics - YouTube

SOLVED: What Is the efficiency of Camnot engine working between 500 K and 850 K? If the engine absorbs 1200 cal of work, how much heat is given to sink? 2 Heat

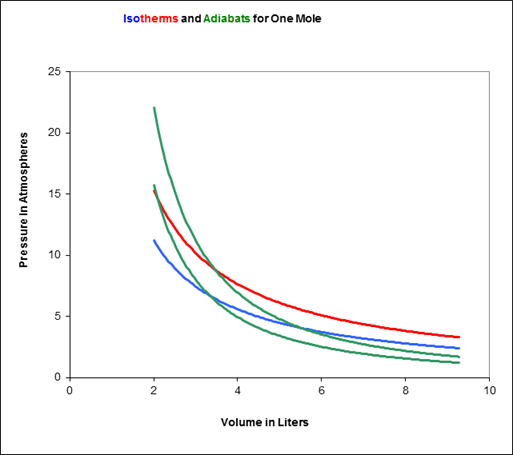
An ideal gas for which Cv = 3R/2 is the working substance of a Carnot engine. During the isothermal expansion the volume doubles. The ratio of the final volume to the initial