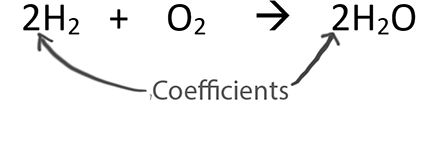Direct formation of hydrogen peroxide from H2/O2 using a gold catalyst - Chemical Communications (RSC Publishing)

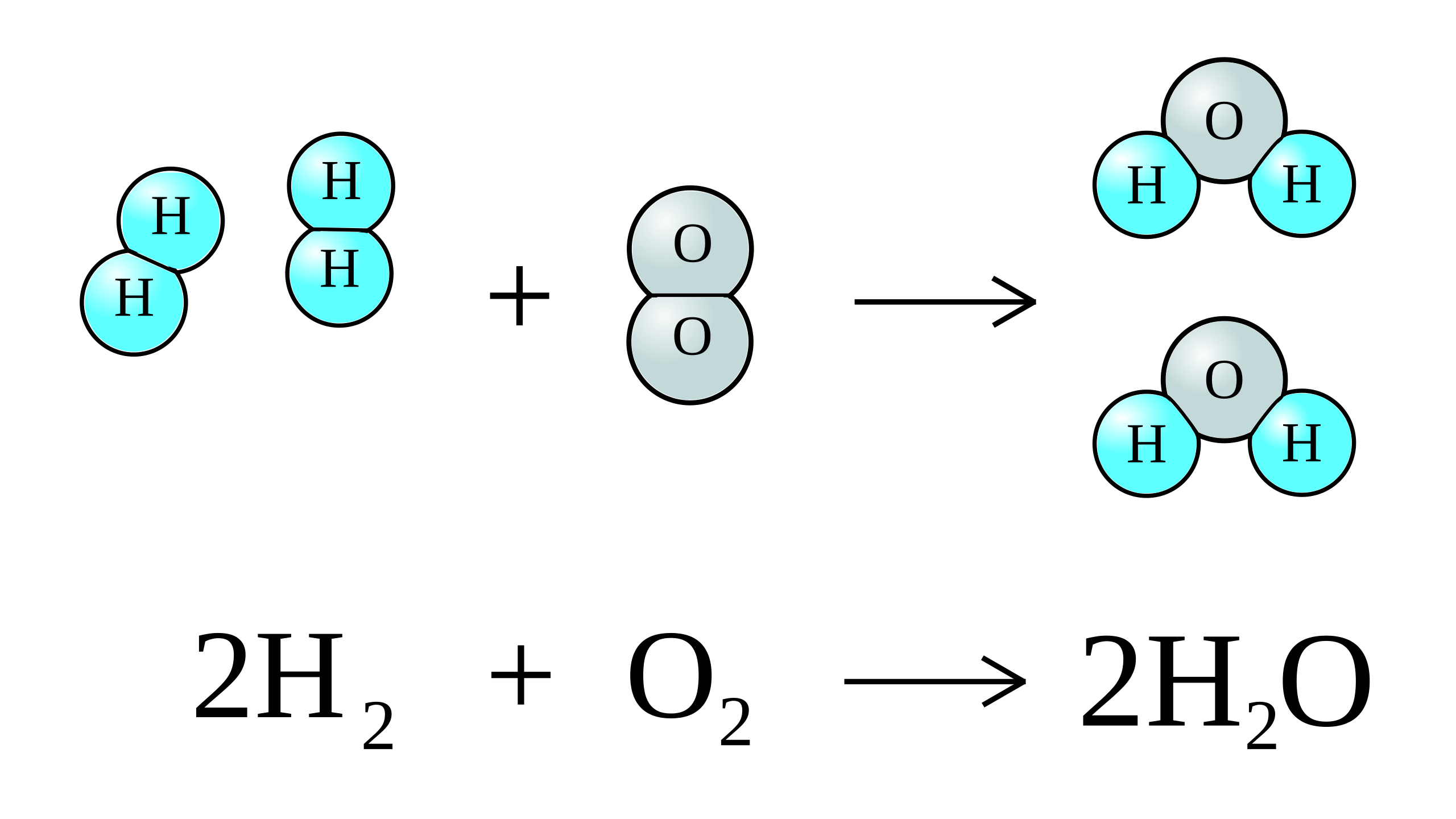
SOLVED: 2. A. Consider the equation, 2H2 + O2 → 2H2O. Here, H2 and O2 are called . Group of answer choices reactants products catalysts modules alkalis B. In chemical equations, the

The H2O2 reaction on polycrystalline Pt electrode during the negative... | Download Scientific Diagram

Two-channel photocatalytic production of H2O2 over g-C3N4 nanosheets modified with perylene imides - ScienceDirect
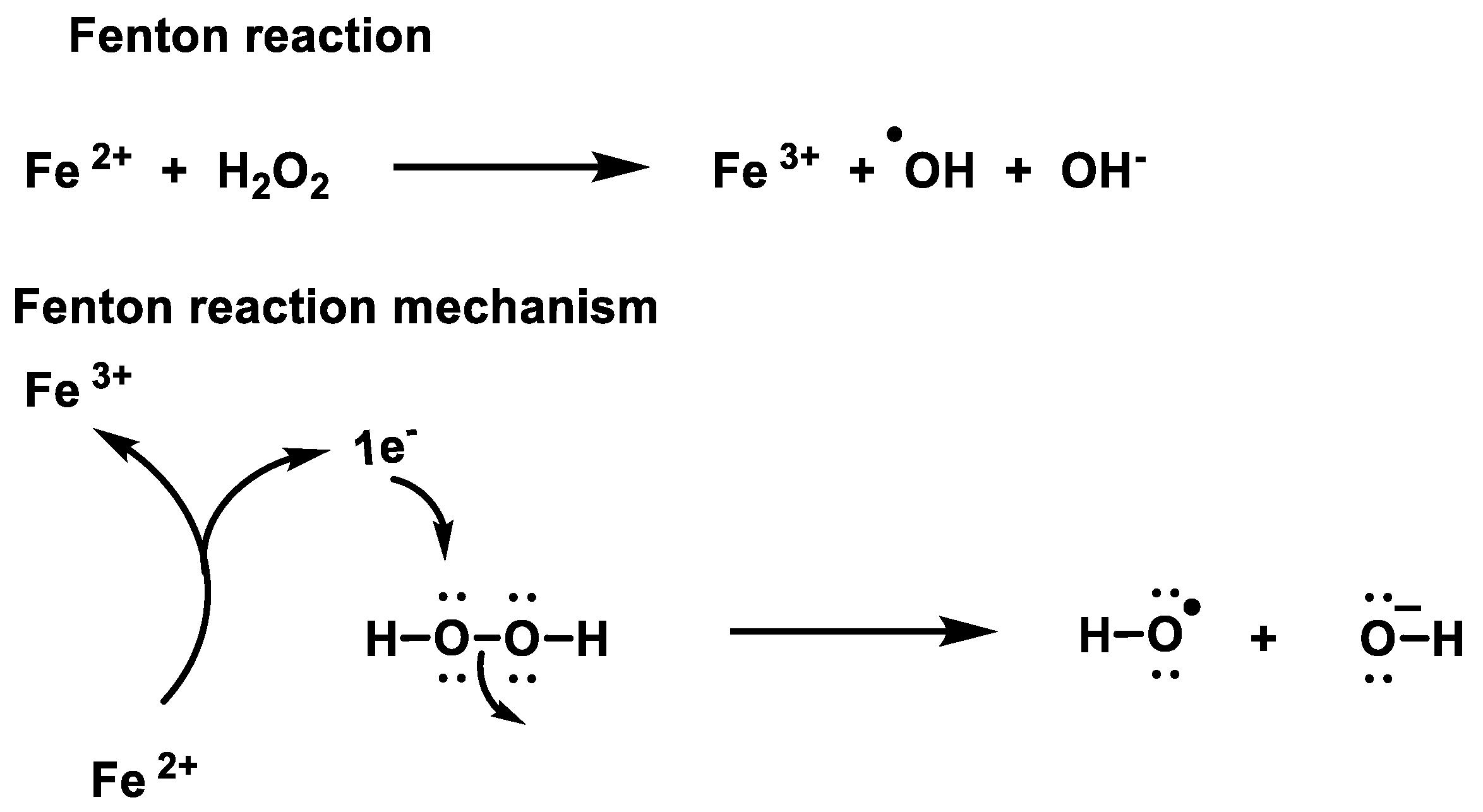
Stresses | Free Full-Text | Chemistry of Hydrogen Peroxide Formation and Elimination in Mammalian Cells, and Its Role in Various Pathologies
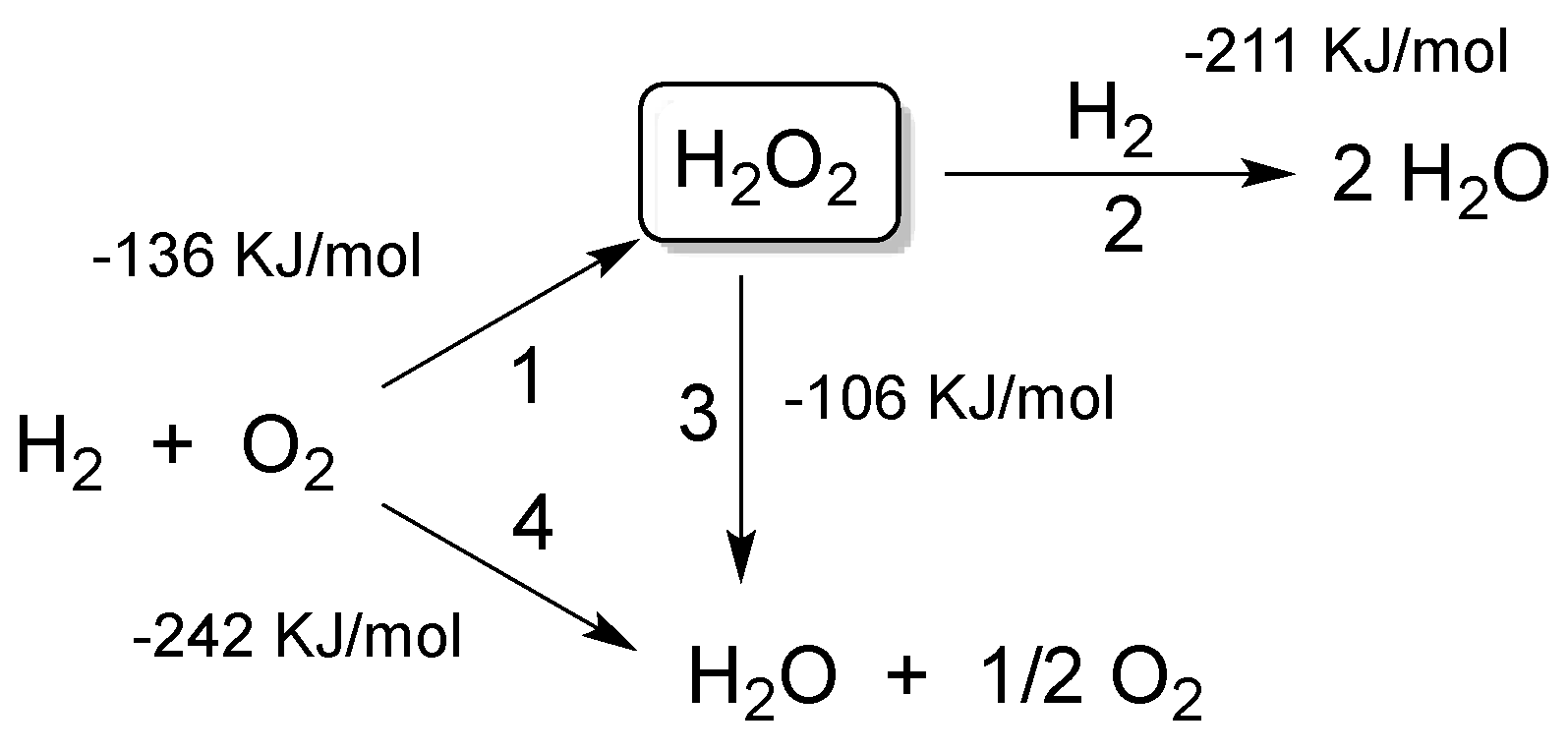
Catalysts | Free Full-Text | Looking for the “Dream Catalyst” for Hydrogen Peroxide Production from Hydrogen and Oxygen

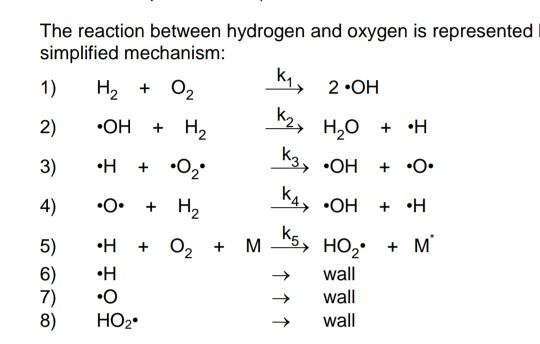
Comprehensive H2/O2 kinetic model for high‐pressure combustion - Burke - 2012 - International Journal of Chemical Kinetics - Wiley Online Library

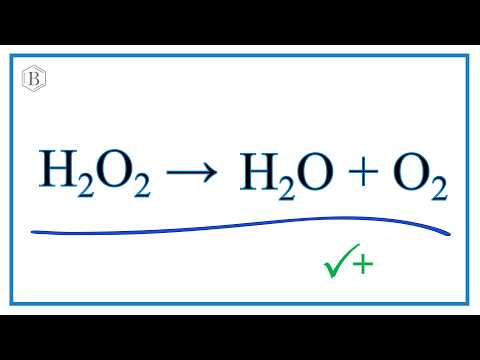
Balance the following chemical equation H2O2 + O3 H2O + O2 Indicating the changes in oxidation numbers of oxygen, find the equivalent weight of H2O2 for this reaction.
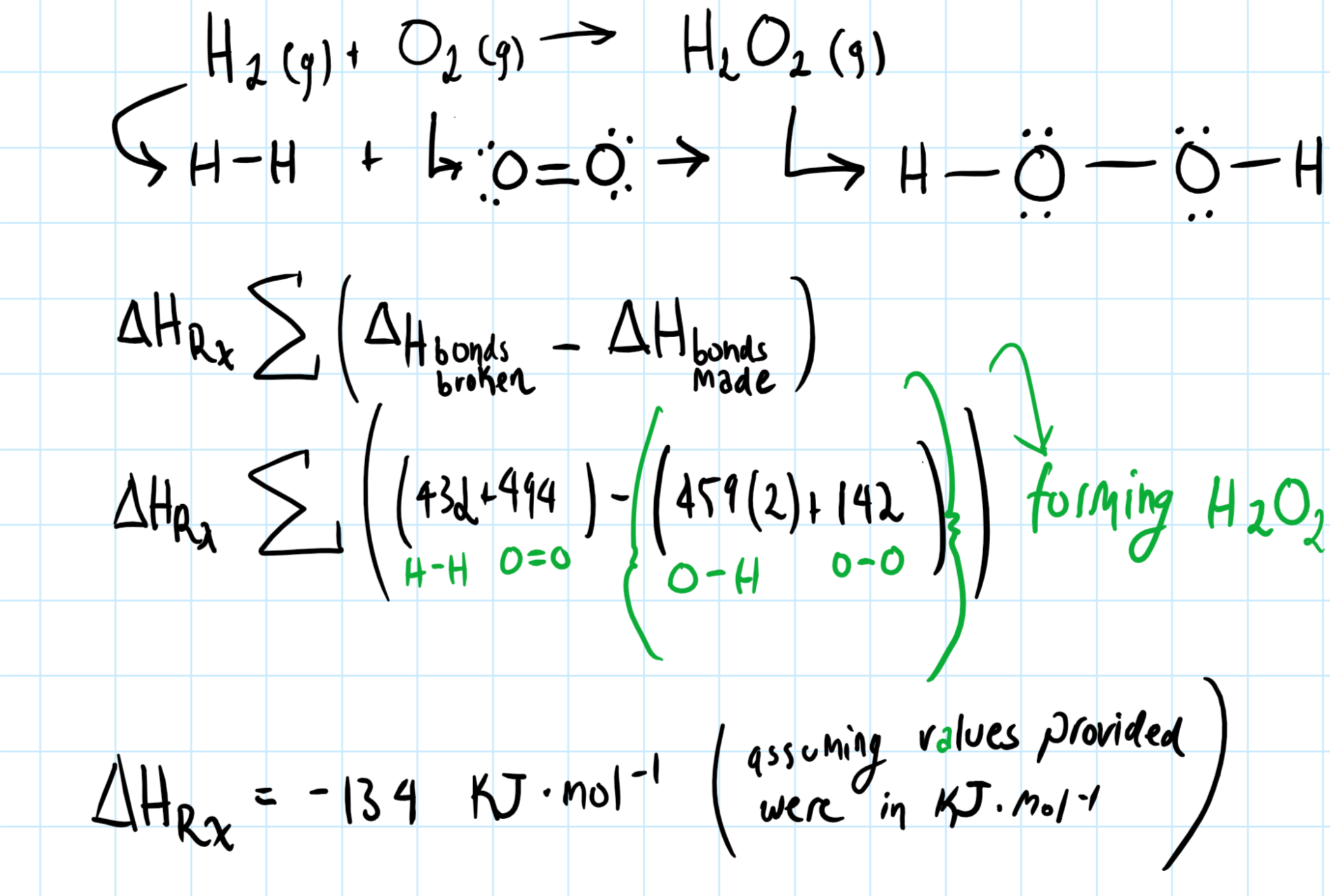
How do you determine the heat of formation of hydrogen peroxide from the given bond energies: H_2(g) + O_2(g) -> H_2O_2(g)? Bond energies: H-H = 432; O2 = 494, O-H = 459. O-O = 142? | Socratic

Enabling Direct H2O2 Production in Acidic Media through Rational Design of Transition Metal Single Atom Catalyst - ScienceDirect

Relationship of H2, O2 and H2O in graph STEP 2: The expected graph is... | Download Scientific Diagram






![PDF] Initiation in H2/O2: Rate constants for H2+O2→H+HO2 at high temperature | Semantic Scholar PDF] Initiation in H2/O2: Rate constants for H2+O2→H+HO2 at high temperature | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/59651c1a79d5be73d45e3b492f6f4396965dd05f/5-Table1-1.png)