Question Video: Finding the Specific Latent Heat of fusion of Water given Its Mass, the Rate of Energy Transfer, and the Time Taken to Change State | Nagwa
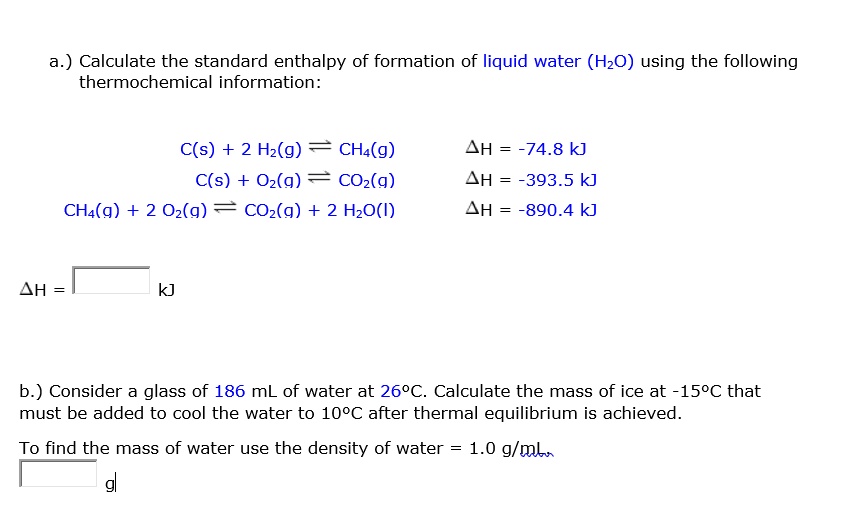
SOLVED: a.) Calculate the standard enthalpy of formation of liquid water (HzO) using the following thermochemical information: C(s) + 2 Hz(g) CHa(g) C(s) Oz(g) COz(g) CHa(g) + 2 Oz(g) COz(g) + 2

The enthalpy of fusion of ice is 6.02kJ mol^-1 . The heat capacity of water is 4.18J(g^oC)^-1 . What is the smallest number of ice cubes at 0^oC , each containing one

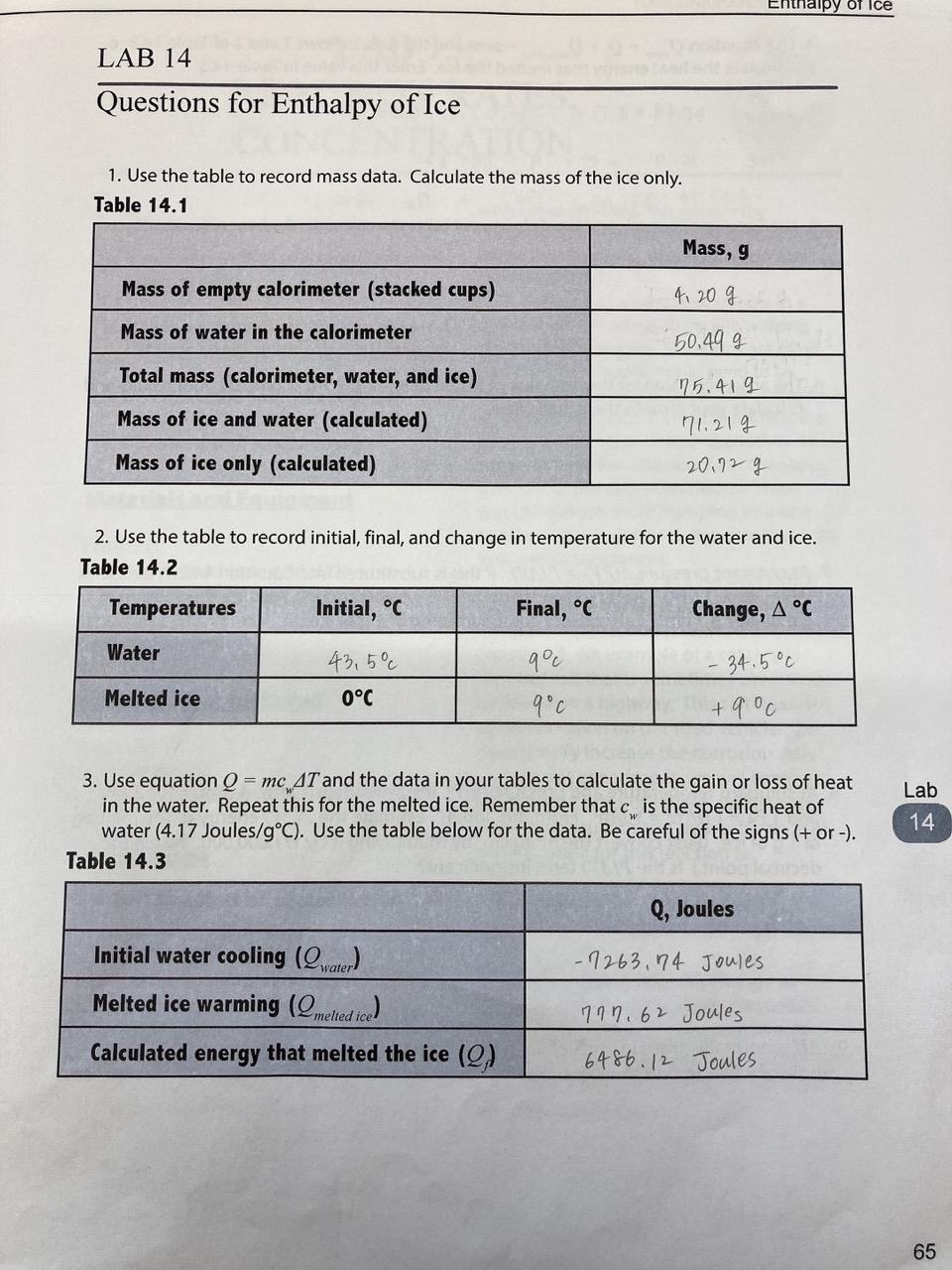
Calculate the heat of fusion of ice from the following data of ice at `0^@C` added to water. Mass of - YouTube




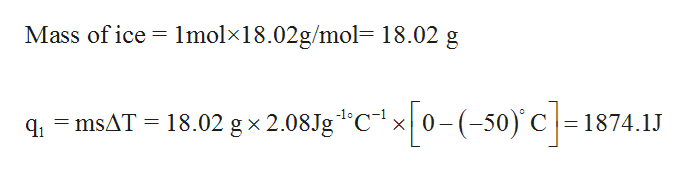

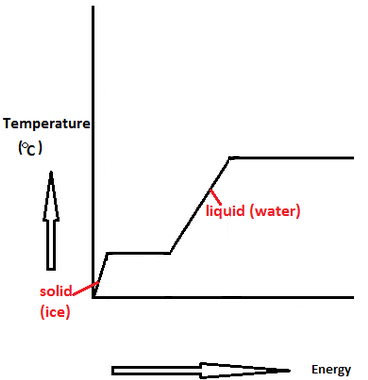
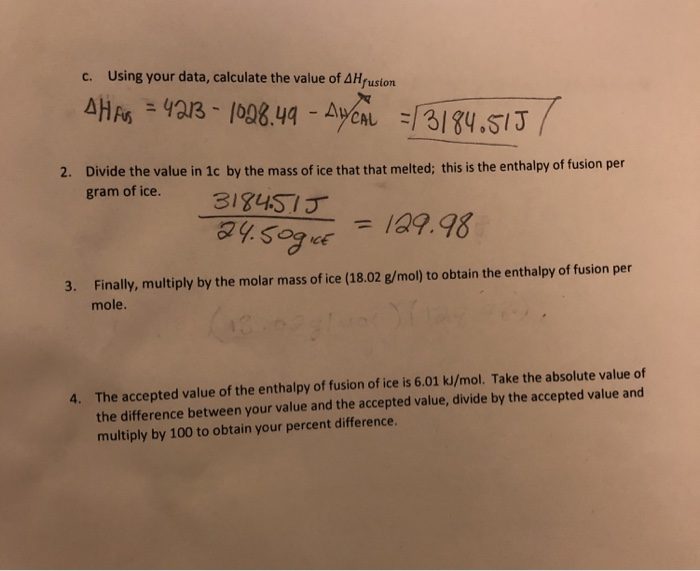



:max_bytes(150000):strip_icc()/GettyImages-1070123348-c9a136e91e7f4b6f9848581aa28b26d1.jpg)