
What is the pH of the solution at half neutralization in the titration of 0.1 N CH3COOH solution by 0.5 M KOH solution? Ka ( CH3COOH = 1.8 × 10^-5 )

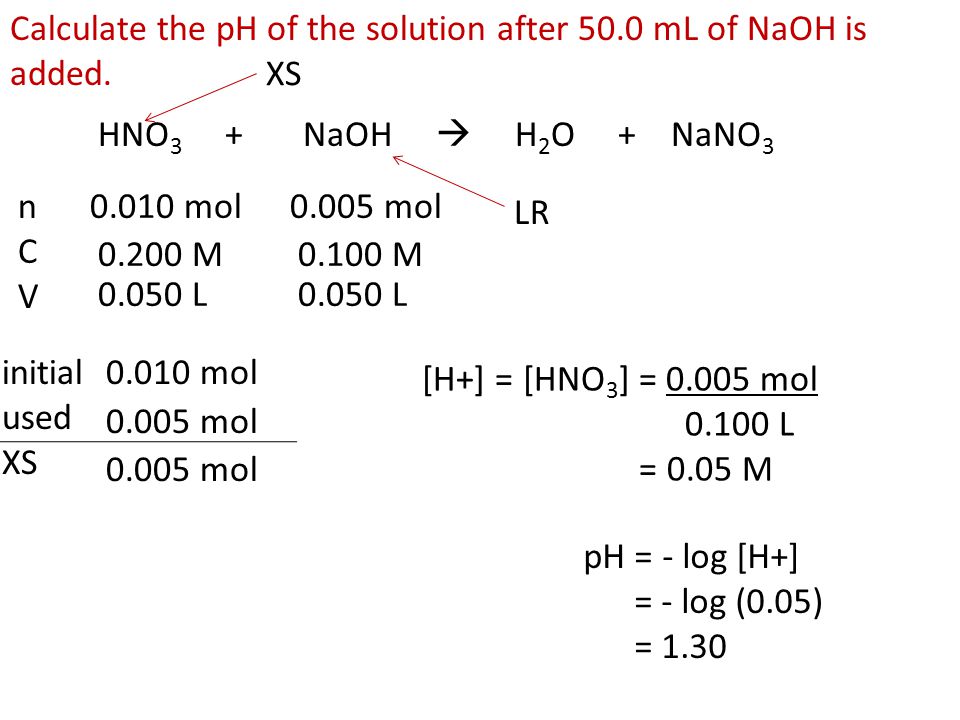
SOLVED: Practice Problem: Calculating pH during Titration Strong Base-Weak Acid ANSWER Calculate the pH of a solution formed when 50.0 mL of 0.100 M CH,COOH is titrated with 45.0 mL of 0.100

SOLVED: 7) You are titrating 100 mL of0.2 MHF with 0.2 M KOH a) Write out the complete, balanced neutralization reaction b) Calculate the pH of the 100 mL solution of 0.2
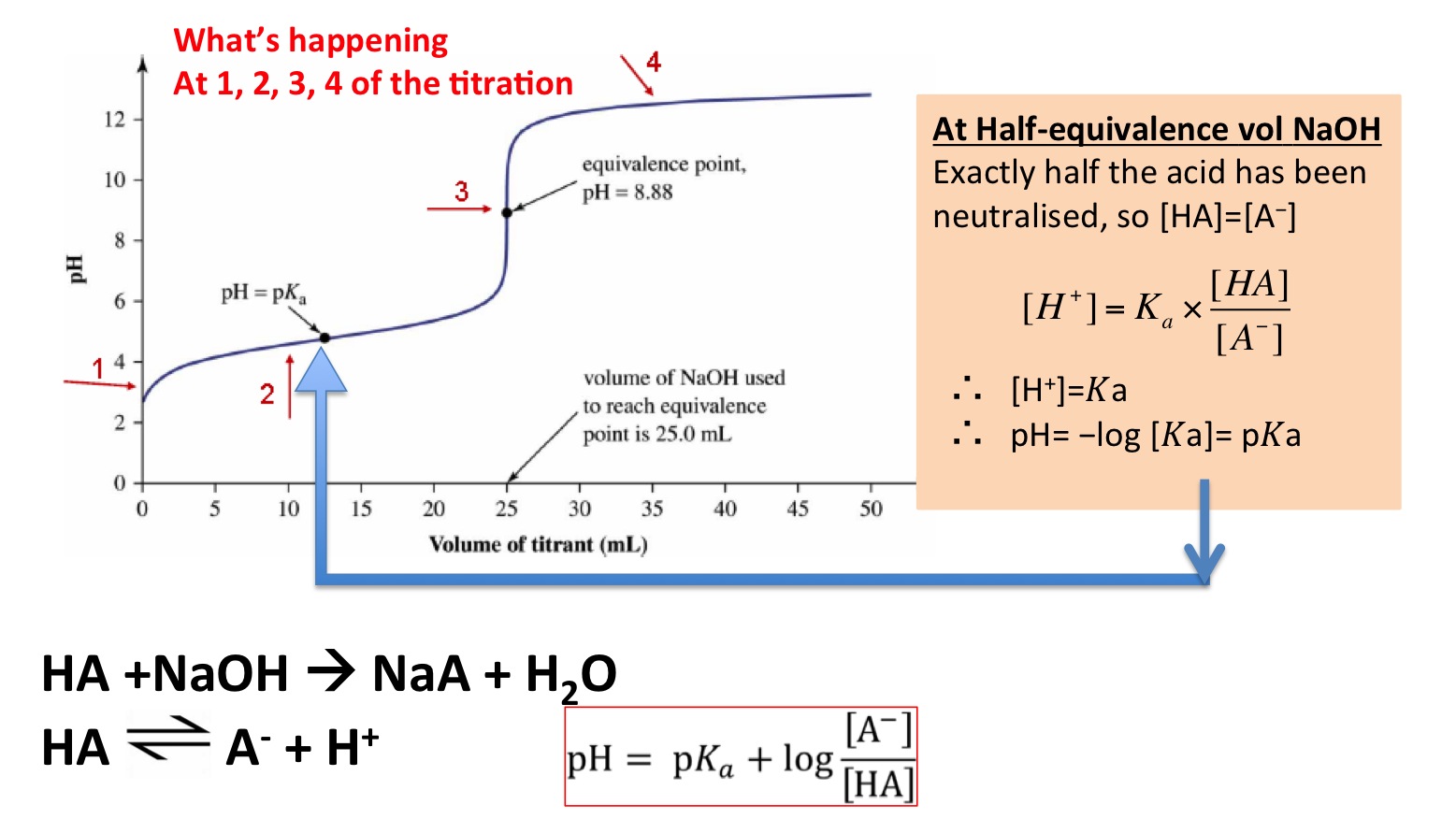
The "pH" at one-half the equivalence point in an acid-base titration was found to be 5.67. What is the value of K_a for this unknown acid? | Socratic

The rapid change of pH near the stoichiometric point of an acid - base titration is the basis of indicator detection. The pH of the solution is related to the ratio of















![Solved Calculate the [H^+], pH, and pOH for the reaction | Chegg.com Solved Calculate the [H^+], pH, and pOH for the reaction | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2Ff7a%2Ff7a2942f-de12-42f0-a39c-6b56694db083%2FphpEUwXKS.png)