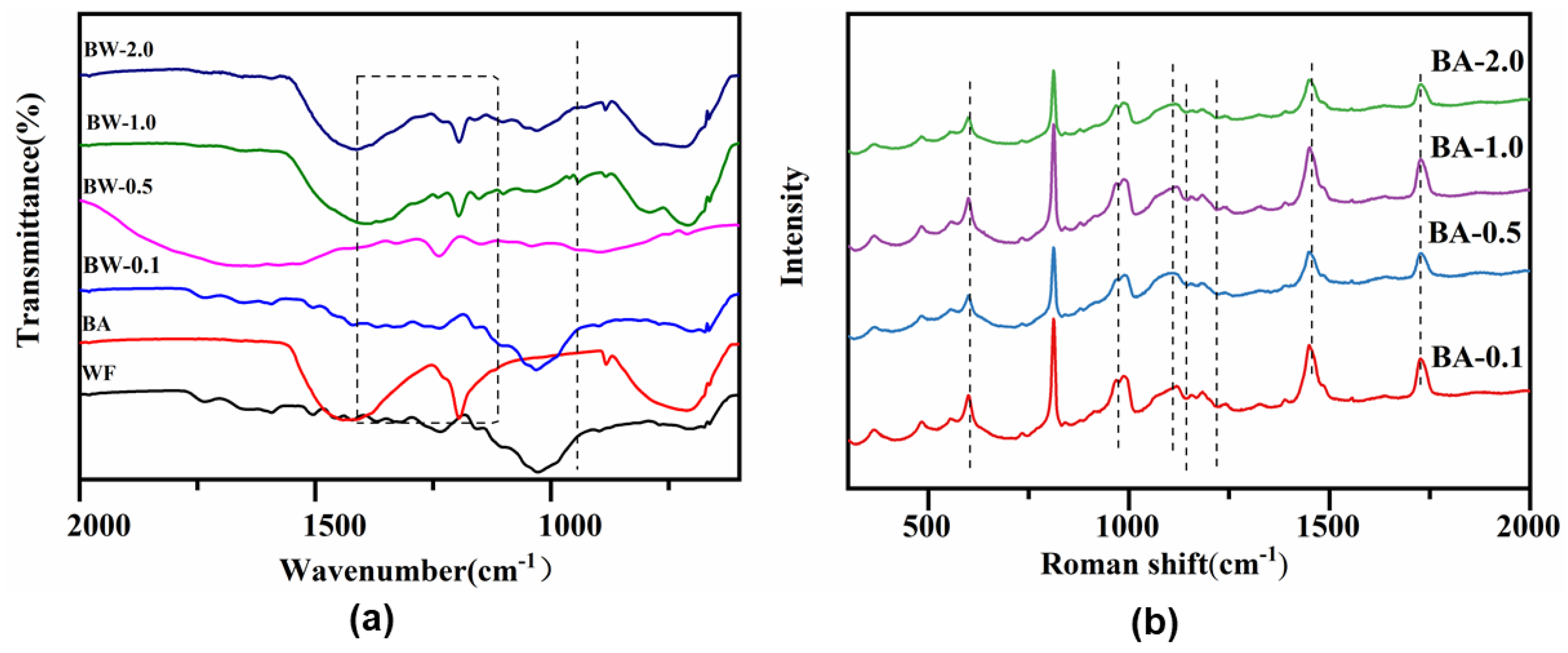
Catalysts | Free Full-Text | Selectively Producing Acetic Acid via Boric Acid-Catalyzed Fast Pyrolysis of Woody Biomass

SOLVED: Boron (B), one of the constituent elements of boric acid, naturally occurs as isotopes of 10B and 11B with percent natural abundance as 20% and 80%, respectively. Boric acid (H3BO3) is

Ultralong Room‐Temperature Phosphorescence from Boric Acid - Zheng - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Optimization of boric acid extraction from ulexite mineral by using supercritical carbon dioxide - ScienceDirect

Variation of pH as a function of boric acid concentration including the... | Download Scientific Diagram
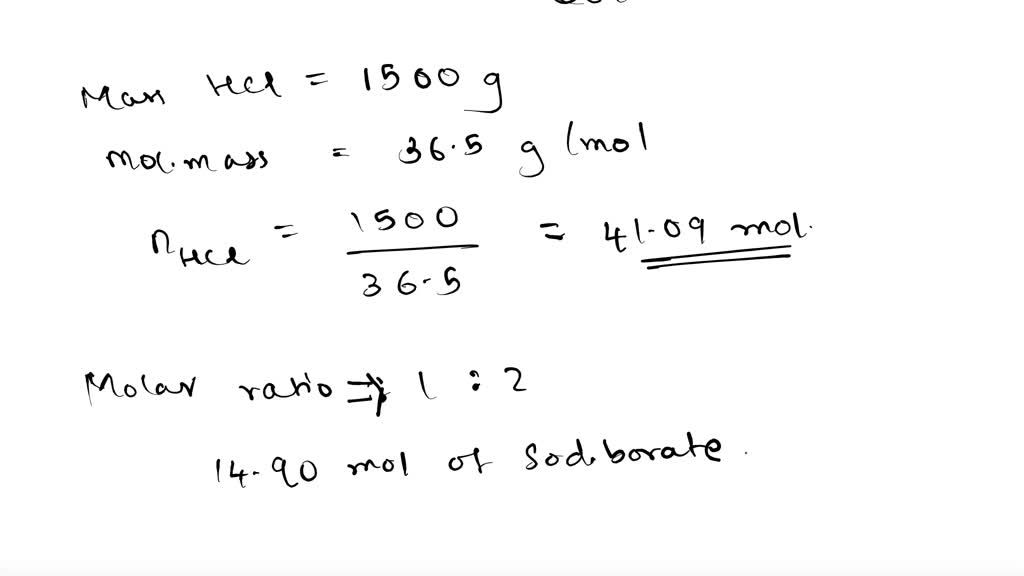
SOLVED: The raw materials, 3 kg of sodium borate along with 1.5 kg of hydrochloric acid will need to produce boric acid. Show the balanced chemical reaction of sodium borate plus hydrochloric

Calculate the mass of 1.00 mmol (S) of trans-cinnamic acid, and the theoretical yield of 2,3-dibromo-3phenylpropanoic acid. | Homework.Study.com
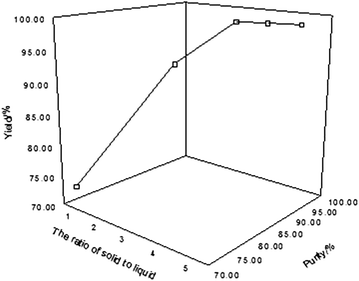
Preparation and characterization of 10B boric acid with high purity for nuclear industry | SpringerPlus | Full Text

A solution of 8% boric acid is to be diluted by adding a 2% boric acid solution to it. The resulting mixture is to be more than 4% but less than 6%

PDF) Novel (Glycerol)borate-Based Ionic Liquids: An Experimental and Theoretical Study | Fabio Bellina - Academia.edu