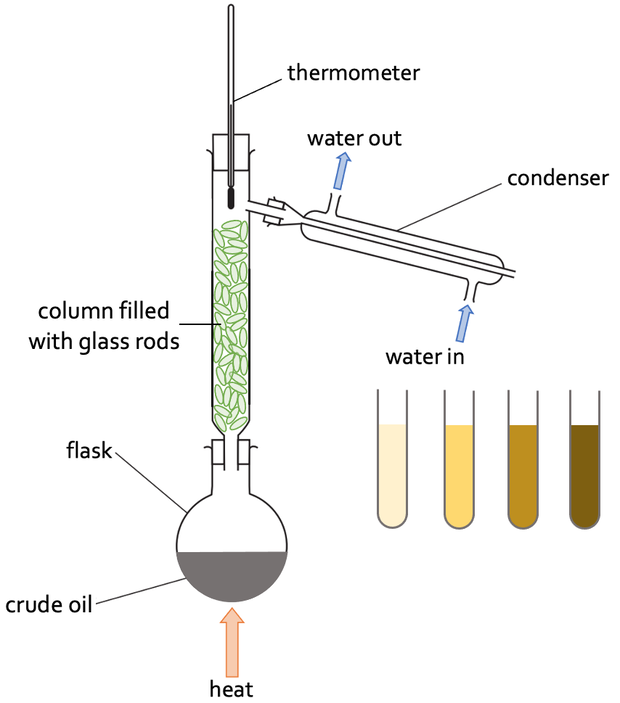
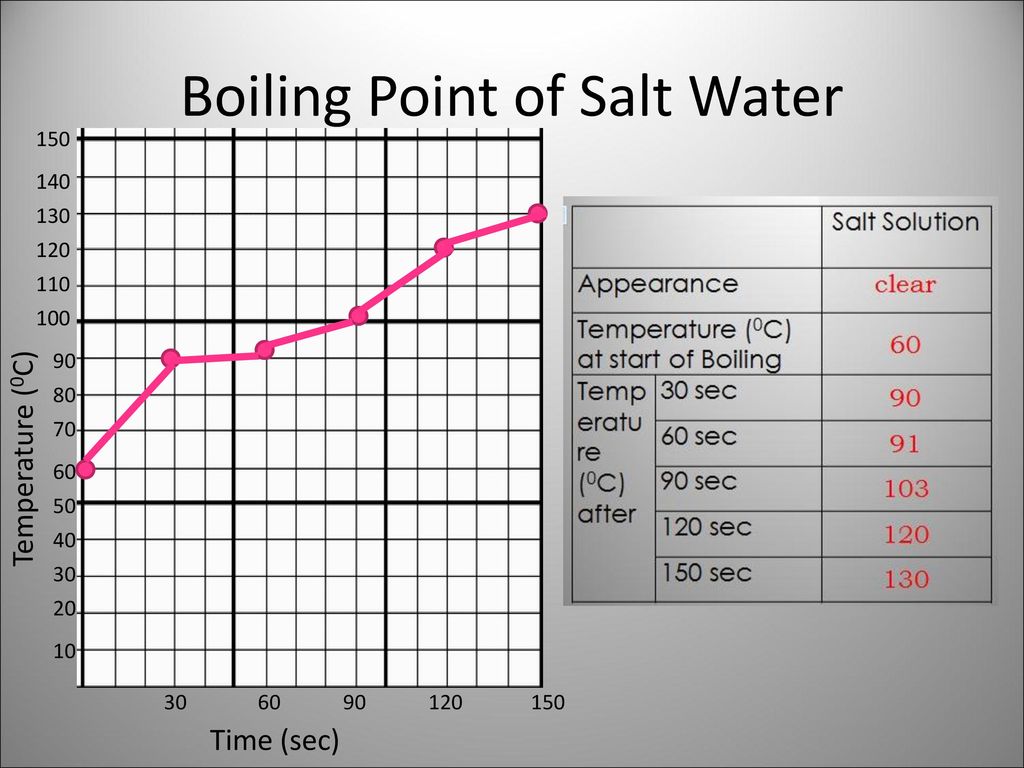
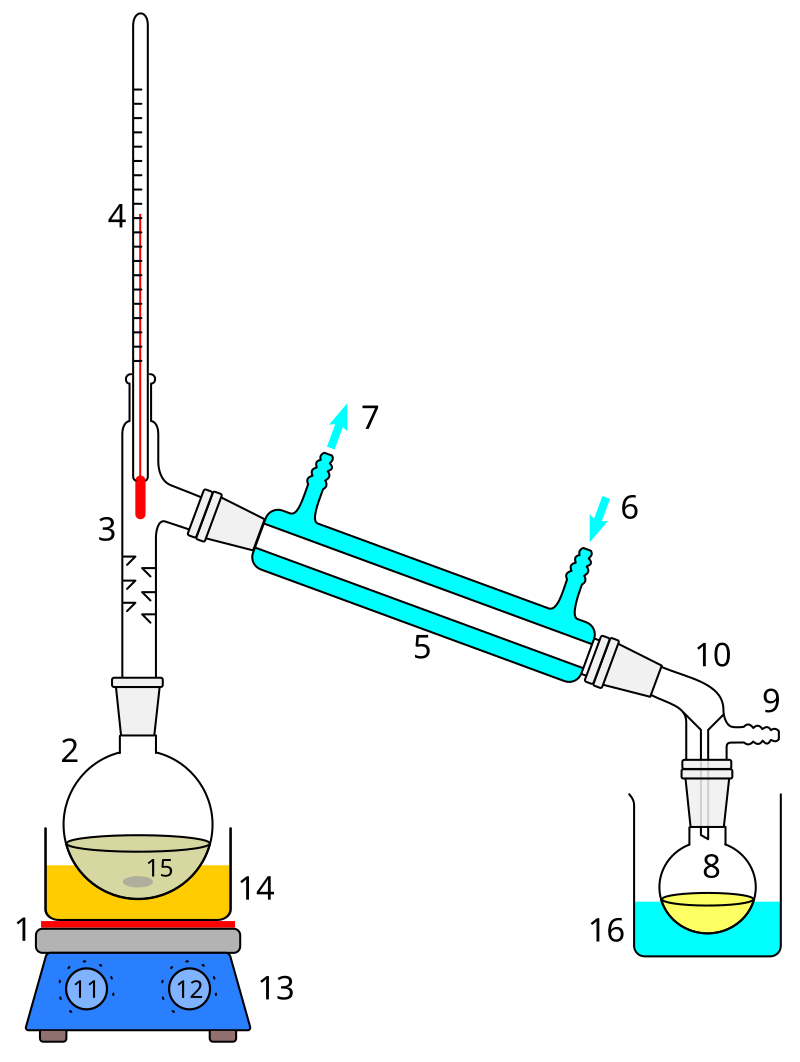
SOLVED: 6. How would the freezing and boiling points of distilled water and seawater compare? Both the freezing and boiling - points of seawater would be lower: Both the freezing and boiling

SOLVED: nigher HINT: freezing Remember, pressure the lowering colligative freezing properties we are studying in Chapter 16.3 are vapor point depression, and boiling-point elevation For each pair of why solutions listed below;
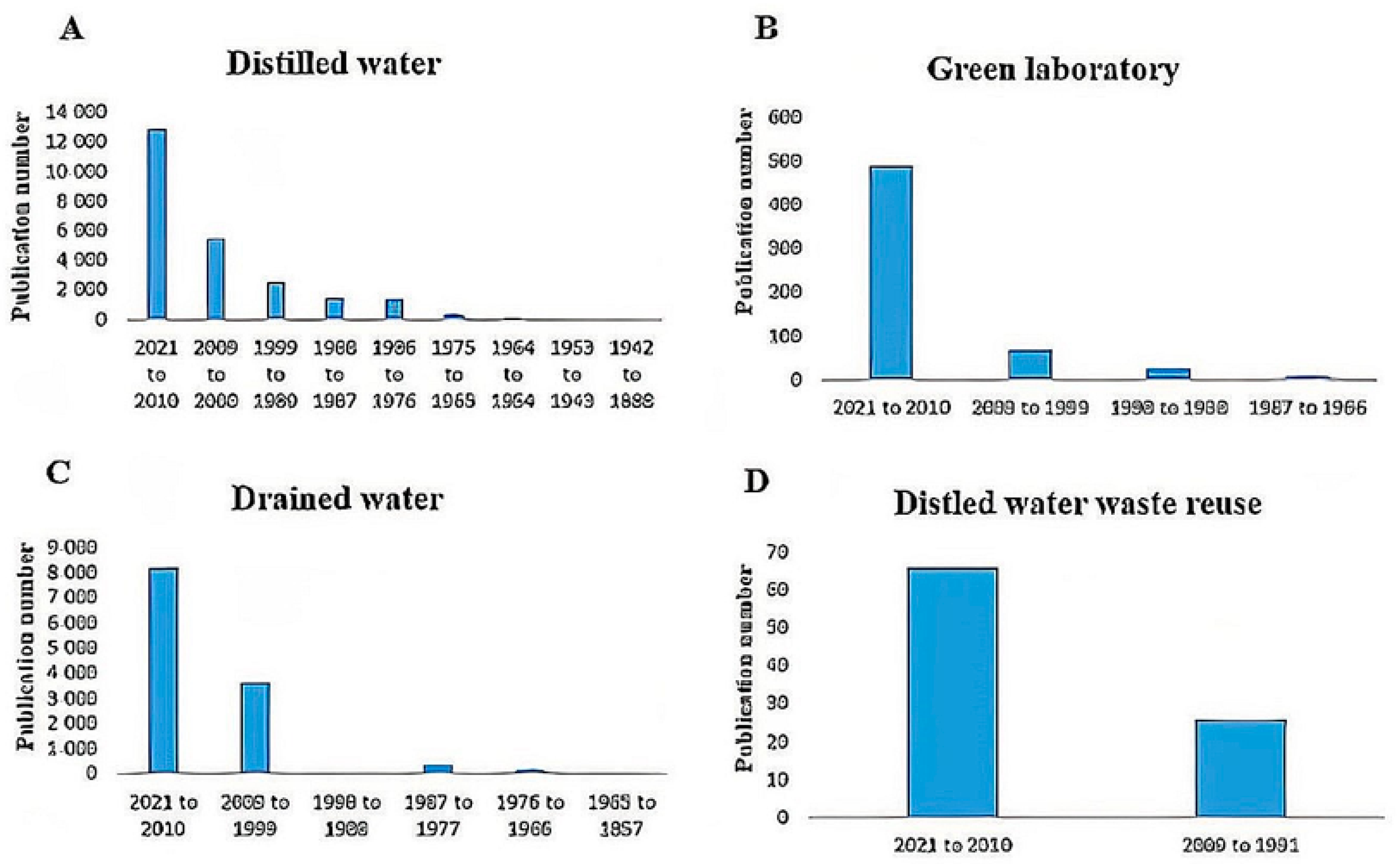
Water | Free Full-Text | Statistical Analysis of Green Laboratory Practice Survey: Conservation on Non-Distilled Water from Distillation Process

SOLVED: 6. How would the freezing and boiling points of distilled water and seawater compare? Both the freezing and boiling - points of seawater would be lower: Both the freezing and boiling