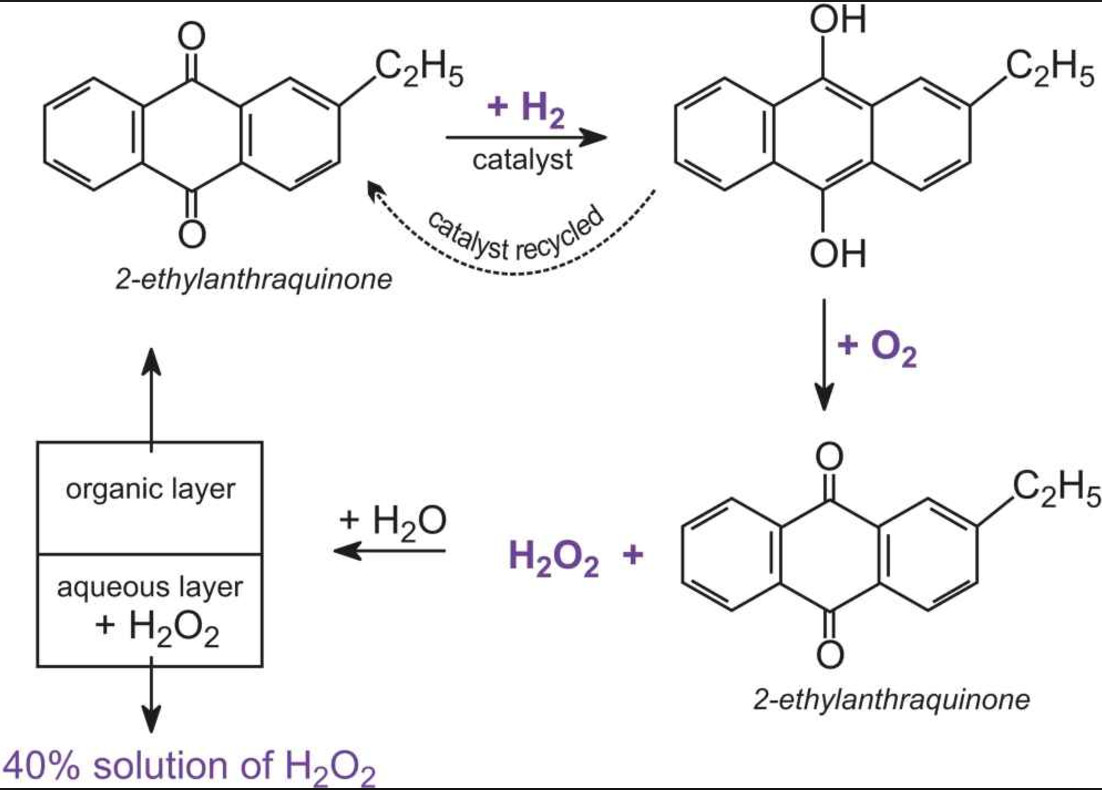
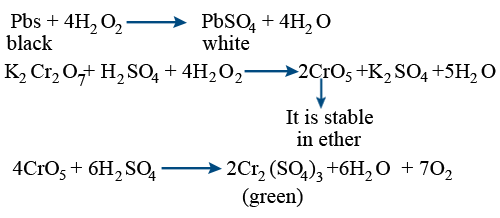
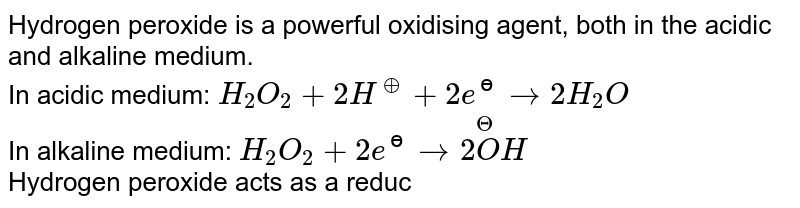While sulphur dioxide and hydrogen peroxide can act as oxidising as well as reducing agents in their reactions, ozone and nitric acid act only as oxidants. Why ?

how can hydrogen peroxide be both oxidising and reducing agent - Chemistry - Redox Reactions - 13207969 | Meritnation.com
Hydrogen peroxide acts as both a reducing agent and oxidizing agent depending upon the nature of the reacting species. In which case does peroxide act as a reducing agent in acid medium? -

Write the chemical reaction to justify that hydrogen peroxide can function as an oxidising as well as - Brainly.in

Relate chemical activity to oxidizing and reducing strength. Explain the concept of disproportionation. - ppt download

An acidic solution of hydrogen peroxide behaves as an oxidising as well as reducing agent. - YouTube
How will you show that H2O2 acts as both oxidising and reducing agent? What is meant by '30 volume' of H2O2? - Quora

While sulphur dioxide and hydrogen peroxide can act as oxidising as well as reducing agents in their - Brainly.in
While sulphur dioxide and hydrogen peroxide can actas oxidising as well as reducing agents in theirreactions, ozone and nitric acid act only as oxidants.This is because(a) in SO2 and H2O2, S and